Californian Liz Reese loves to hang ten, but chronic shoulder pain kept her out of the waves. A total shoulder replacement with Dr. Ari Youderian got her back on her board.
Exactech Active Intelligence Technology
Using the Latest Advancements to Improve Joint Replacement Surgery Outcomes

Looking for a doctor who uses Active Intelligence technology?
Take the next step.
Search by specialty and city, state or zip to find a surgeon near you.
Exactech ActiveIntelligence Technology
For many years, surgeons have used X-rays, surgical tools, and special techniques to plan surgeries. But now, joint replacement technology can help your surgeon plan, prepare and execute your surgery with a goal of better outcomes.
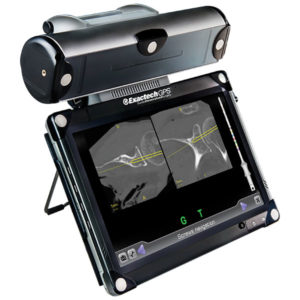
You may have heard of robotics, robot-assisted surgery or computer-assisted surgery. Exactech Active Intelligence surgeons use a computer-assisted surgical technology called ExactechGPS Guided Personalized Surgery.
ExactechGPS Guided Personalized Surgery
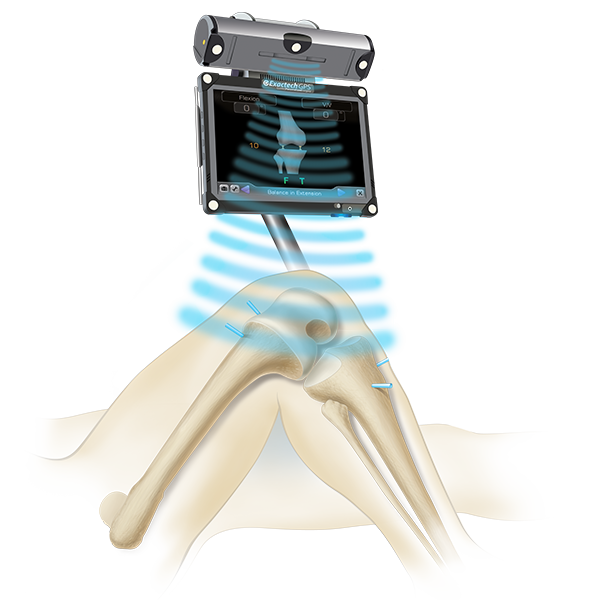
To get the best results for your joint replacement surgery, your surgeon must understand YOUR unique anatomy so they can accurately place your implant. The better aligned your implant is, the better your results will be.
ExactechGPS is a computer-assisted tool that gives surgeons a 3D view of your joint. This allows your surgeon to make small changes and place your implant just where it should be.
How ExactechGPS Technology Works:
If your knee implant is placed even as little as 3 degrees off, it can increase the risk of your knee replacement not working like it should.1
ExactechGPS has helped surgeons place knee replacement implants less than 1 degree from the perfect position.2
Watch the video below to see how ExactechGPS Knee Technology Works.

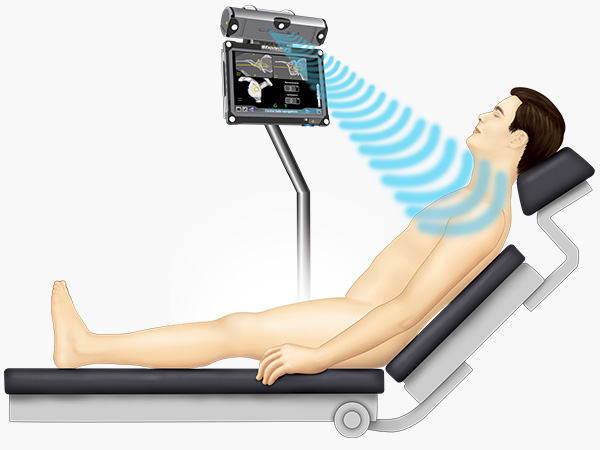
ExactechGPS Shoulder Applications help surgeons plan for your surgery using a virtual simulation on a computer.
Prior to surgery, Active Intelligence surgeons use your CT scan to create a 3D model on a computer to clearly see your shoulder joint and bone structure. This helps the surgeon decide where to make bone cuts and place your implant.
Personalized just for you, your surgeon can plan your surgery through a virtual simulation before even entering the operating room. Then, during surgery, your doctor will use this plan to place your shoulder implant just where it should be.1
Watch the video below to see how ExactechGPS Shoulder Technology Works.
Patient Stories
Video: Rita, ExactechGPS Knee Replacement Patient Story
Knee pain was getting in the way of Rita enjoying retirement. After replacing both her knees with ExactechGPS, she is getting her life back.
Video: Jerry ExactechGPS Knee Patient Story
Aircraft mechanic Jerry Smith was experiencing constant knee pain as a result of a broken leg earlier in life.
Video: Barbara, ExactechGPS Knee Patient Story
Total knee replacement surgery with ExactechGPS helped Barbara get back to teaching aerobic exercise at her local senior center.
Video: Jackie, ExactechGPS Shoulder Replacement Patient Story
Video: Martha, ExactechGPS Shoulder Replacement Patient Story
Bothersome shoulder pain was preventing Martha from spending precious time with her grandchildren. With the help of Dr. Byram and ExactechGPS, she’s back to what she loves.
Answers to the most frequently asked questions.
Accurate positioning of your knee or shoulder replacement is very important to the success and longevity of the implant. Computer-assisted technology can give your surgeon greater visibility of your joint while helping more precisely place your implant.
In a survey of potential joint replacement patients, 87% of those considering joint replacement surgery said they prefer computer-assisted surgery over robotics. Many respondents said they are more comfortable knowing the surgeon’s hands are performing the surgery and not a robot.2
When it comes to knee replacement surgery using ExactechGPS, the short answer is no. Following a surgeon’s initial learning curve with new technology, the average operating time using ExactechGPS is proven to be similar to that of conventional knee replacement surgery.2
Resources for ExactechGPS Guided Personalized Surgery
Looking for a doctor who uses Active Intelligence technology?
Take the next step.
Search by specialty and city, state or zip to find a surgeon near you.
With any surgery, there are potential risks, and results will vary depending on the patient. Joint replacement surgery is not for everyone. Check with your physician to determine if you are a candidate for joint replacement surgery. Your physician will consider the risks and benefits associated with this procedure, as well as individual factors such as the cause of your condition, your age, height, weight and activity level.
The information contained within this website is for educational purposes only and is not providing medical advice. This information is not intended to replace the expert guidance of your orthopaedic surgeon. Please direct any questions or concerns you may have to your orthopaedic surgeon. Decisions concerning patient care and treatment should be made solely by your physician(s). With any surgery, there are potential risks and results will vary depending on the patient.
- Ritter MA, Faris PM, Keating EM, Meding JB. Postoperative alignment of total knee replacement: its effect on survival. Clin Orthop Relat Res. 1994; 299:153-156.
- Data on file at Exactech.*
*In vitro (bench) test results may not necessarily be indicative of clinical performance
ExactechGPS® is manufactured by Blue Ortho® and distributed by Exactech.